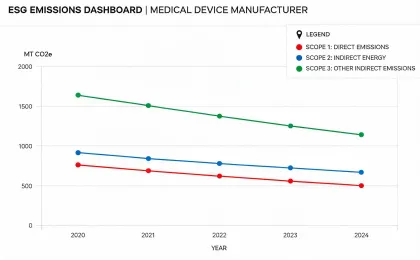
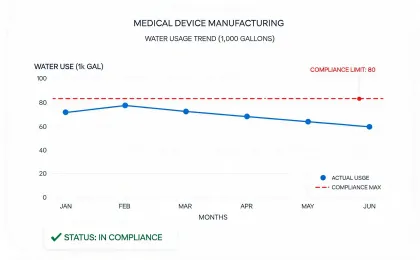
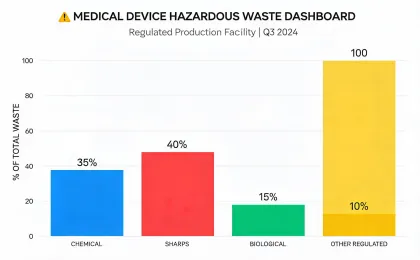
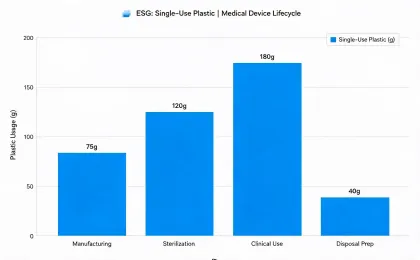
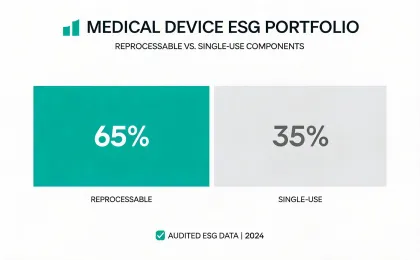
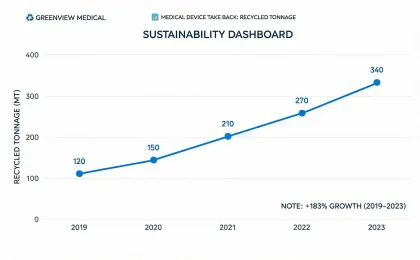
Reporting is organized around medical equipment operations, service logistics, waste handling, device longevity, and climate-related disclosures that procurement and finance teams can compare.






Globus Medical sustainability files are written for reviewers who need substance: material declarations, packaging reduction notes, service-life assumptions, take-back logistics, supplier screening, and the operational impact of remote support. Each report is tied to a facility planning decision, so ESG work can support procurement instead of becoming a detached annual statement.
Technical documentation references packaging, materials, and end-of-life handling where relevant to EU medical device submissions.
Critical suppliers are reviewed for energy source, logistics distance, regulated waste controls, and audit responsiveness.
Remote triage reduces unnecessary truck rolls while preserving service documentation, cybersecurity logs, and escalation transparency.